Open Cooling systems
In many industrial manufacturing processes, water as cooling agent is an essential auxiliary substance, which is mostly available in sufficient quantities and at low costs. Due to the excellent thermodynamic properties and the environmentally highly beneficial application this „natural“ raw material is excellently suitable for the cooling down also of larger heat sources.
To be able to eliminate large amounts of heat, which cannot be used technically in any other way,
mostly „open cooling systems“ with an evaporative cooling towers are used. The heated cooling water is sprayed against the counter current airflow in the cooling towers. The cooling down of the cooling water is achieved by withdrawal of the evaporative heat and convection with air.
However, in practice there are three technical problems regarding the application of (commercial) water as cooling agent
- Corrosive effect of water for metals
- Formation of lime/salt deposits at the heat exchanging surfaces
- Microbial growth at installations and walls

EFFECTIVE OPERATION
An essential precondition for an efficient and flawless operation of „open cooling systems“ are maximum of operating safety and functionality. Since the composition and the quality of the used water mostly vary from plant to plant, an individual water treatment must be carried out to allow a hygienic and economical operation of the plant.
Due to the operating conditions in the cooling towers, there is principally a corrosive effect of the cooling water for the iron material. Furthermore, also nonferrous metals, such as copper and brass, are affected. Here, an appropriate material selection is of the same importance as a consequent monitoring and control of the operating water. Only the combination will allow a long-term maintenance of the plant value. For example, it may be necessary to add corrosion inhibitors to the make-up water during application. These will form a protective film on the material surfaces and will thus reduce the possibilities of effect for the corrosive water.

In addition to the corrosive effect, the components of the operating water must be considered: Salt deposits, especially lime formation, originate from the water evaporation and the continuous supply of non-softened or only partially softened additional water into the cooling circulation. The concentration of dissolved salts and also of dissolved lime in the commercial water increases and in this way results in a formation of deposits, if the solubility limits are exceeded.
For an economical operation, i.e. low fresh water supply and economically optimized condition of the plant at the same time, raising the solubility limits by addition of small quantities of stabilizers has demonstrated good results for many years. Here, especially the hardness stabilizer must be added continuously and mass-proportionally to the make-up water.
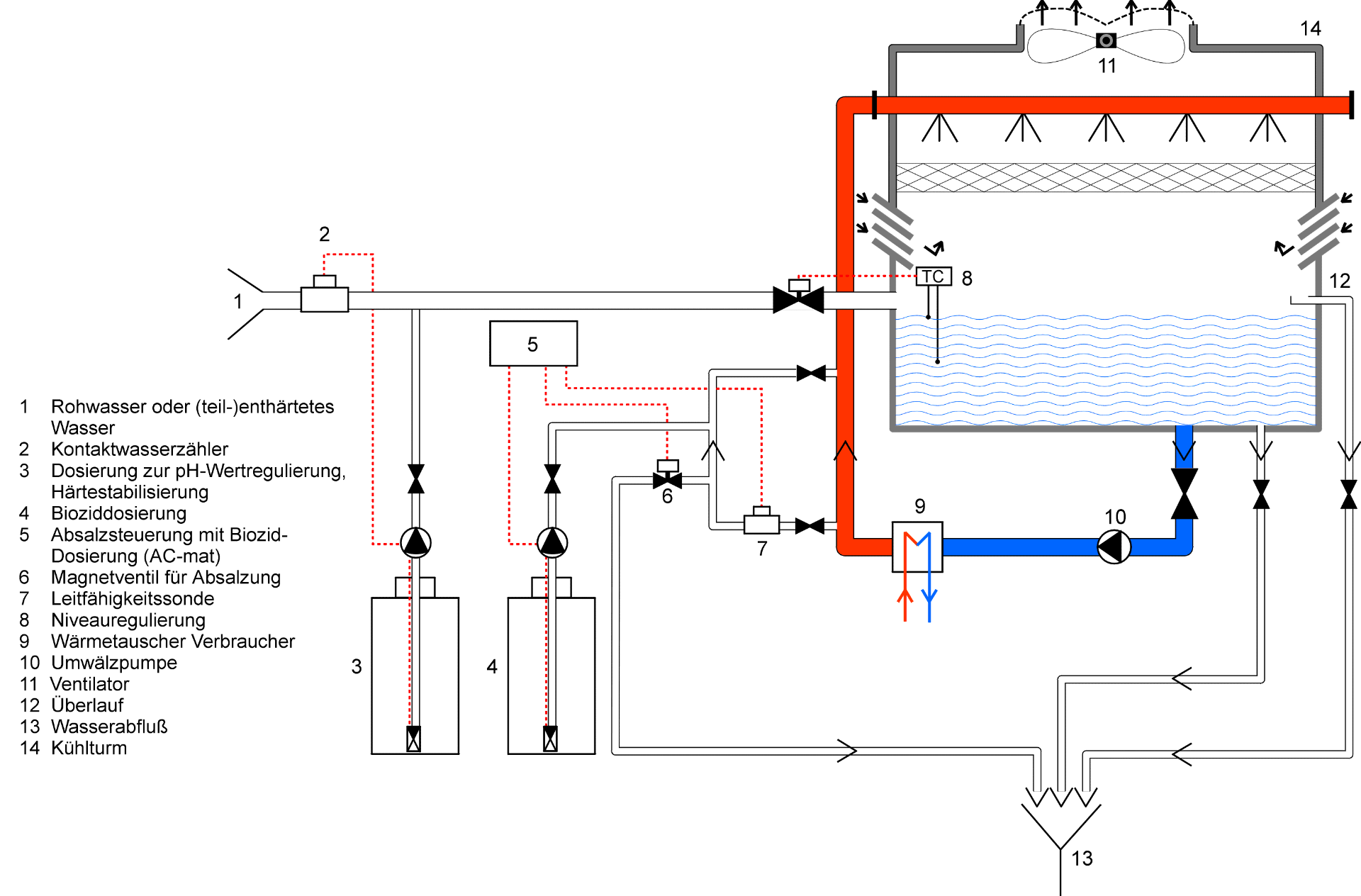
A frequently observed problem in „open cooling systems“ is microbial growth, especially in evaporative cooling towers. Algae, bacteria and fungi may proliferate rapidly, because high nutrient concentrations due to addition of organic material and a favourable water temperature create practically ideal living conditions:
Algae show by a green colour of the water and form alga sheets. Bacteria form unsightly mucilaginous layers on the water surface and cause a thick vegetation on the walls and
installations. Especially at the heat exchanging surfaces these layers as well as the calcareous deposits are highly undesirable.
Also, the metabolic products of the bacteria contained in the deposits might cause a corrosion of the metal surfaces (pitting). In addition, fungi may affect the wooden components in the cooling towers and destroy them. Vital disadvantages for the operation of cooling plants originate, if the heat exchange is impaired by deposit formation at the heat exchanging surfaces, since the listed up organic as well as inorganic substances act like insulators.
Impurities and deposit formations drastically increase the power consumption
Experiments show that already with a 0.1 mm mucous layer the heat transition is reduced by about 5 – 8 %. With a mucous layer thickness of 1 mm, the cooling performance is further decreased, so that the degree of efficacy may be reduced by approximately 20 - 25 %. A comparable loss in performance is caused by calcareous deposits at the heat exchanging surfaces. In these cases, the cooling water flow must be increased to eliminate the same amount of heat.
As a consequence, a considerably higher pump performance and thus more power consumption are required.
Deposit formation and pipe clogging are frequent causes for unexpected function impairment of the connected heat sources, i.e. the production has to be stopped, if the heat of the machines is not sufficiently eliminated.
In addition, the removal of alga sheets, mucous bacteria layers, fungi and lime always requires intensive cleaning and should therefore be avoided in any case.
To optimize the heat transition at the heat exchanging surfaces and for an energy saving operation, a consistent water treatment in the „open coolant circuits“ is just as important as the maintenance of the primary manufacturing plants.
How does lime scale originate?
The undesired formation of calcareous deposits (scale) originates due to the interaction of the temperature and chemical equilibrium reactions.
(I) CO2 + H2O H2CO3
(II) H2CO3 + H2O H3O+ + HCO3
(III) 2 HCO3- CO32- + CO2 + H2O
(IV) Ca2+ + CO32- CaCO3
The equations (I) and (II) are reactions where due to dissolution of CO2 in water carbon dioxide and hydrogen carbonate are formed. Due to the contact of the circuit water with the atmosphere and a temperature increase, the dissolved CO2 escapes and more and more carbonate is formed because of the shift of the chemical equilibrium (equation (III)) to the right. The higher the water temperature, the more CO2 will escape, because the gas solubility decreases. As soon as a high carbonate concentration exists and the soluble product of calcite is exceeded (corresponding to exceeding of the lime-carbon dioxide equilibrium), scale precipitates from the water to form disturbing deposits, e.g. at the heat exchangers.
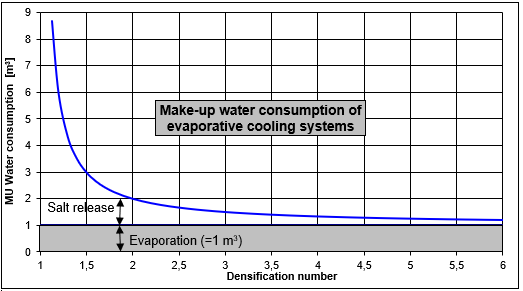
Correlation of thickening and water consumption
As is shown in the below figure, the water consumption can be considerably reduced by targeted thickening of the salts contained in the cooling water. A remarkable aspect is the great difference between the saving effect of 1.2 X and 3 X thickening. In contrast, thickening above 4 results in only minor additional water saving and mostly includes corrosion problems. Our specialists will determine the optimum thickening number for your system and will help you to reduce your water costs.
Your advantages with a consistent water treatment in „open cooling systems“ are:
- Corrosion protection through formation of a protective layer on the metal surfaces
- Prevention of calcareous deposits with hardness stabilizers and controlled water exchange („salt release“)
- Considerably improved water quality through reduced bacterial and algal burden
- Improved air quality of the environment through reduced odor and germ burden
- Improved appearance of the cooling towers
- Reduced costs through reduced cleaning requirements
- Energy saving potential in water costs through reduced fresh and waste water
- Maintenance of the plant value through corrosion protection
- Energy saving through prevention of calcareous and mucous deposit formation at the heat exchanging surfaces
Example of the maintenance program for open cooling systems
- Carry out a one-time initial cleaning with specially designed chemicals
- Revise make-up water preparation (eg softening)
- Apply a suitably selected corrosion inhibitor and / or hardness stabilizer depending on the amount of make-up water
- Dispense the appropriate biocide and control the so-called water hygiene
- Regularly monitor the physical-chemical parameters of the circuit (pH, conductivity, content of active ingredients,…)


